|
 CAS: 59-05-2 CAS: 59-05-2
Structure:
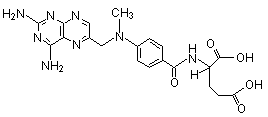
 Specifications: Conforms to standard USP 27 and EP4,BP2002 Specifications: Conforms to standard USP 27 and EP4,BP2002
| Text |
USP27 |
EP4 |
BP2002 |
| Assay% |
98.0-102.0 |
98.0-102.0 |
98.0-102.0 |
| Residue on ignition% |
≤0.1 |
|
|
| Specific roataion |
+19-+24 |
+19-+24 |
|
| Loss on drying% |
|
|
|
| Chromatographic purity% |
≤2.0(total) |
|
|
| |
≤0.5(individual) |
|
|
| Water% |
≤12.0 |
≤13.0 |
≤12.0 |
| Organic Volatile impurites |
conforms |
| Sulphated ash% |
|
≤0.1 |
≤0.1 |
| Heavy metals ppm |
|
50 |
1 |
| Related substance |
|
|
|
| Total |
|
≤1.3 |
≤1.3% |
| Individual |
|
At most five peaks |
At most five peaks |
| Impurity A% |
|
≤0.5 |
≤0.5 |
| Impurity B% |
|
≤0.5 |
≤0.5 |
| Impurity C% |
|
≤0.5 |
≤0.5 |
| Impurity D% |
|
≤0.5 |
≤0.5 |
| Impurity E% |
|
≤0.5 |
≤0.5 |
| Unknown Impurity % |
|
≤0.5 |
≤0.5 |
| (R)-Methotrexate% |
|
≤3.0 |
≤3.0 |
 Physical properties: Physical properties:
Appearance: orange yellow crystal powders
Solubility: the production hardly dissolves in water, ethanol, chloroform or ether, and it easily dissolves in sparse alkali, and it can dissolve in dilute hydrochloric acid.
 USE: The product is used as anti-tumour medicament. USE: The product is used as anti-tumour medicament.
 Package: It's packed 2-ply polyethylene bags are heat sealed, Net weight 1KG/Tin. Package: It's packed 2-ply polyethylene bags are heat sealed, Net weight 1KG/Tin.
|

